Medical device risk management template5/24/2023 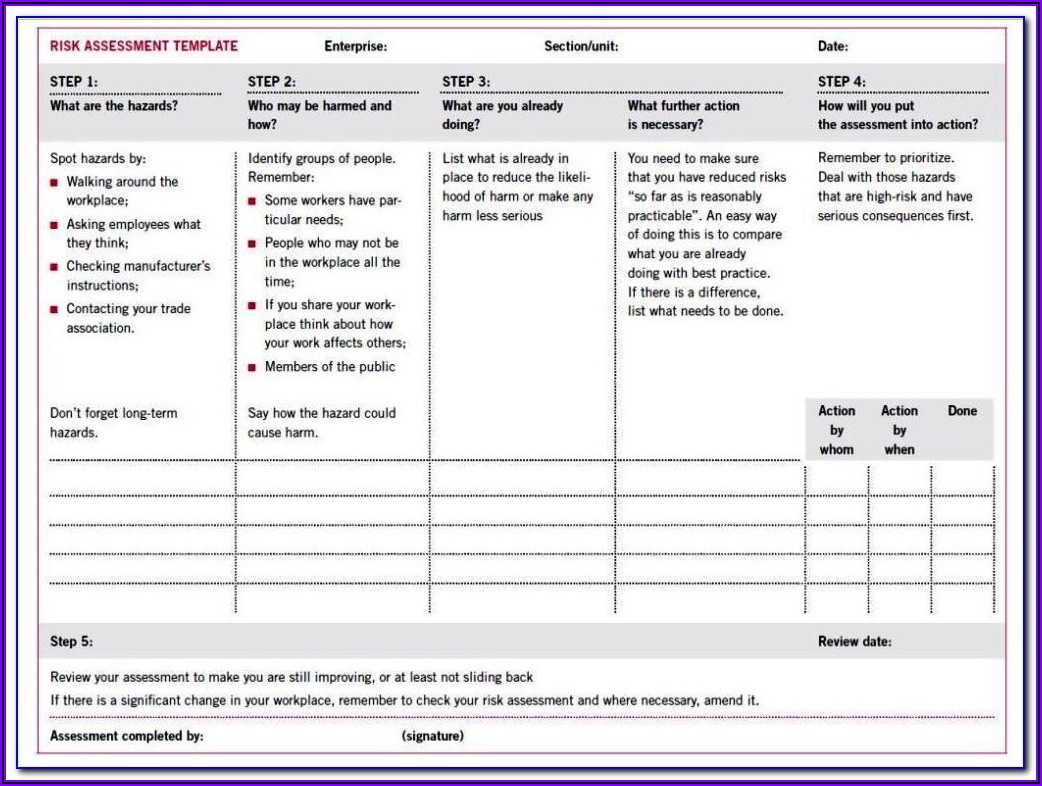
Estimate and evaluate the risks associated with, and occurring during, the intended use and during reasonably foreseeable misuse.Identify the known and foreseeable hazards associated with the device.Establish and document a risk management plan for each device.Manufacturers, under MDR, must implement the following aspects of risk management to be fully compliant. Risk Management under MDRĪnnex I section 3 of EU MDR 2017/745 mentions the risk management requirements specific to the European medical device regulations. According to the definition, risk management strategies help prevent particular harm or risk and prevent severe harm. Medical Device Risk- DefinitionĪs per Article II of EU MDR 2017/745, medical device risk is defined as ‘the combination of the probability of occurrence of harm and the severity of that harm’. These strategies may either eliminate the risk or mitigate the overall severity of the risk. Manufacturers are expected to plan, document, and implement risk management strategies in this process. Under EU MDR 2017/745, risk management is a continuous and iterative process. 
It is imperative to understand the medical device’s specific risks to a patient. All medical devices are associated with inherent risks of some level.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
